TTAB Reverses 2(e)(1) Mere Descriptiveness Refusal of "PATCH BOOSTER" for Heart Devices
The Board reversed a Section 2(e)(1) refusal of the mark PATCH BOOSTER, finding it not merely descriptive of permanent and temporary heart assist devices, percutaneous access devices consisting of a disk implanted beneath the skin, implanted pumps, and external drive units therefor. In re L.Vad Technology, Inc., Serial No. 78285714 (April 28, 2006) [not citable].
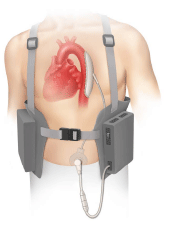
The Examining Attorney advanced three different arguments in support of her assertion that PATCH is merely descriptive of the goods, but all were in vein. [TTABlog pun!]. First, she argued that Applicant's goods act like a "patch" because they mend and strengthen the ability of the heart to function; she pointed to Applicant's statement that its goods "in a sense act as a patch in that they help to keep the patient's heart working." Not a bad argument, right? Wrong. The Board agreed with Applicant L.Vad that "its goods can be deemed to be a patch only figuratively, not literally."
Second, the PTO relied on dictionary definitions and NEXIS excerpts that used the word "patch" in the context of covering a hole in the heart and mending damaged tissue. The Board, however, found that these references do not support a finding that "patch" is merely descriptive because they do not involve tissue patches of the type mentioned in these articles.
Thirdly, the PTO argued that "patch" may be a "drug-impregnated material" worn on the skin, and thus the word describes the "percutaneous access devices" of the application. The Board was not persuaded. Applicant's disk is beneath the skin, not adhered to the skin. "There is no evidence that medication delivery devices implanted beneath the skin are called 'patches.'"
In sum, the Board found that the word PATCH is "at most suggestive of the goods."
As to the word BOOSTER, the Examining Attorney relied on dictionary evidence indicating that "booster" means "something which improves or increases," and she contended that Applicant's goods "are used to boost the ability of the heart to work." The Board found the significance of the word "booster" to be "too vague and generalized to be deemed merely descriptive." Again, the Board found this term to be merely suggestive.
As to the combination PATCH BOOSTER, that too is suggestive, not merely descriptive:
"'Patch booster' would seem to have two possible grammatically correct constructions, neither of which appears to make sense as applied to applicant's identified goods. Whether the composite is viewed to mean 'a patch which boosts,' or is viewed to mean 'a booster for a patch,' the construction is awkward and does not directly describe applicant's goods, which do not involve a 'patch' as that term is defined in the record (i.e., a heart tissue patch or a medication skin patch)."
The Board acknowledged four published instances of use of the term "patch booster," but found that evidence unpersuasive. "These are isolated instances of use spread over thirty-five years" and the paucity of this evidence "weighs against a finding of mere descriptiveness." Moreover, this evidence has "little or no probative value" as to how the term would be perceived today.
Finally, the Board noted that any doubt as to mere descriptiveness must be resolved in Applicant's favor.
In a brief concurring opinion, Judge Bucher noted that "those knowledgeable about the goods will understand the etymology of each individual word." However, "even if the word PATCH conveys information about the nature of the implanted balloon pump portion of the device, and even if the word BOOSTER conveys information about the function of the entire device," when these two arguably descriptive words are combined, the composite "appears to be nothing more than suggestive" of the goods.
Text Copyright John L. Welch 2006.




0 Comments:
Post a Comment
<< Home